3 Key Questions For Sulfur Application
It’s no secret that more and more corn growers are utilizing — and reaping the benefits from — a balanced approach to crop nutrition. In fact, while planning next year’s fertilizer program for your customers, it’s not only imperative to help them get the most out of their fertilizer dollar, it’s also crucial to help today’s corn growers have on hand all the required nutrients available to the corn crop at the right time. Sulfur (S) is an important nutrient to remember to include in a balanced fertility plan.
To ensure investment in S produces more bushels of corn, three key questions must be addressed:
- How much S does corn need and when does it need it?
- What form does S need to be in to be available for uptake by the corn crop?
- How can S be supplied in the right form at the right time?
How much S does corn need and when does it need it?
Sulfur is an essential nutrient for corn growth, and a critical nutrient to make required proteins. Total uptake of S in a corn crop ranges from 0.1 to 0.12 pound S per bushel of corn produced. While this pales in comparison to the total nitrogen (N) required by a corn crop, that same crop cannot maximize its yield potential unless S is available at the right time in non-limiting concentrations.
When is the right time? S uptake occurs over the entire growing season, with relatively constant uptake from the 14-leaf stage to maturity. Unlike nitrogen, in which 70% to 75% of the total N is taken up by flowering, only 40% to 50% of the total S is taken up by flowering (see chart below). Furthermore, S is also immobile within the plant, which means the plant is unable to compensate for low levels of S that may occur late in the season by moving S from older growth to new growth. Therefore, if a steady supply of S is unavailable over the entire season, late-season S deficiencies can significantly impact yield, since S already in the plant cannot be moved for use in the growth of new leaves and grain.
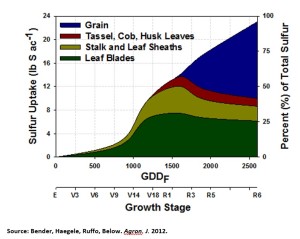
Research has shown that the timing of S uptake by corn is very consistent, even when different varieties and locations are considered. (click to enlarge)
What form does sulfur need to be in to be available for uptake by the corn crop?
In order for S to be available for the corn crop to use, it must be in the form of sulfate (SO4). There are two common forms of S available in fertilizers today: One is in the available form of SO4, and the other is in elemental form. Elemental sulfur (ES) must be broken down by microbes in the soil (oxidized) into SO4 before it is available for uptake by plants. The amount of SO4 released from ES depends on the physical size of the ES; larger pieces take longer to break down than smaller pieces. (Think how much quicker ice cubes melt than a large block of ice; the smaller the particle sizes, the quicker the ES releases.) The environmental conditions also affect how quickly ES converts into SO4. Since microbes dominate the process, warm, moist soils with good aeration result in more ES being released. The oxidation of ES into SO4 means that S is slowly available over time from ES sources.
How can S be supplied in the right form at the right time?
If S must be in SO4 form, why not just put down S in its available form of SO4? Similar to N, S in its available form is subject to losses. Just as we focus on ways of controlling losses of N from the system, S needs to be protected as well. Putting down all of a crop’s S requirements in the available form at one time does not ensure that there is a steady supply all season long. Using a fertilizer that supplies S in both its readily available SO4 form and in a slowly available ES form means that as soon as the fertilizer is put down, there is S available for the crop; and as the growing season progresses, then the ES will be released, ensuring a season-long supply of S that matches crop demand.
As growers continue to expect more yield from high-producing hybrids, it will be necessary to help them fine-tune their crop nutrition plans. As with other nutrients, application, timing and rate of S will be an important piece of the corn yield puzzle.





